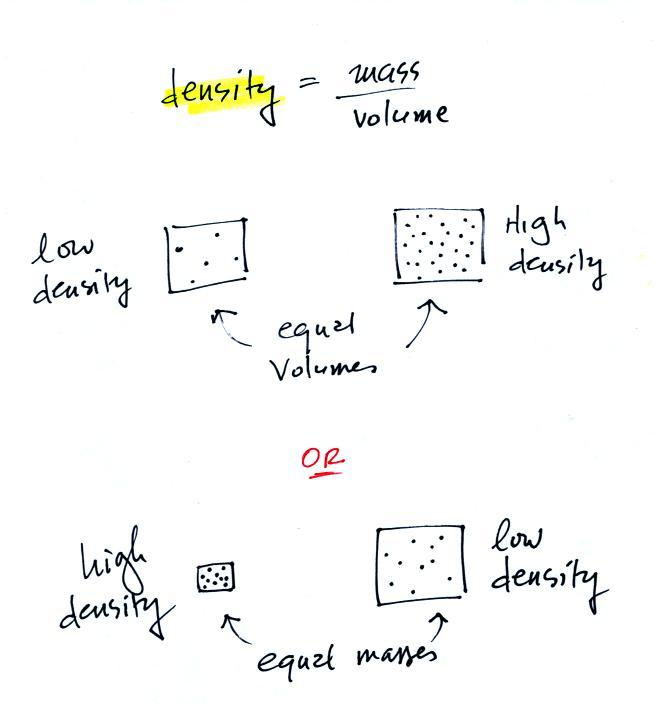
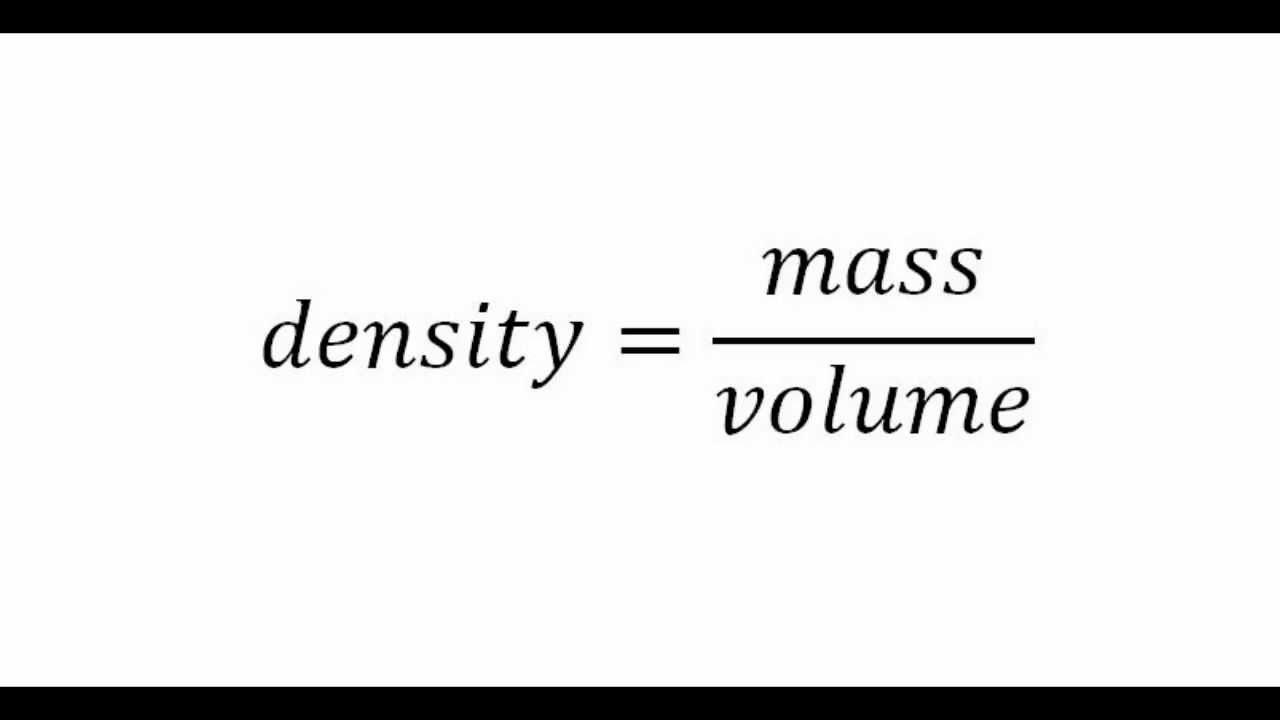Lab/hw) so if the formula is density equals mass over volume, so assume I would subtract mass of empty beaker with mass with unknown liquid and then continue with the formula or
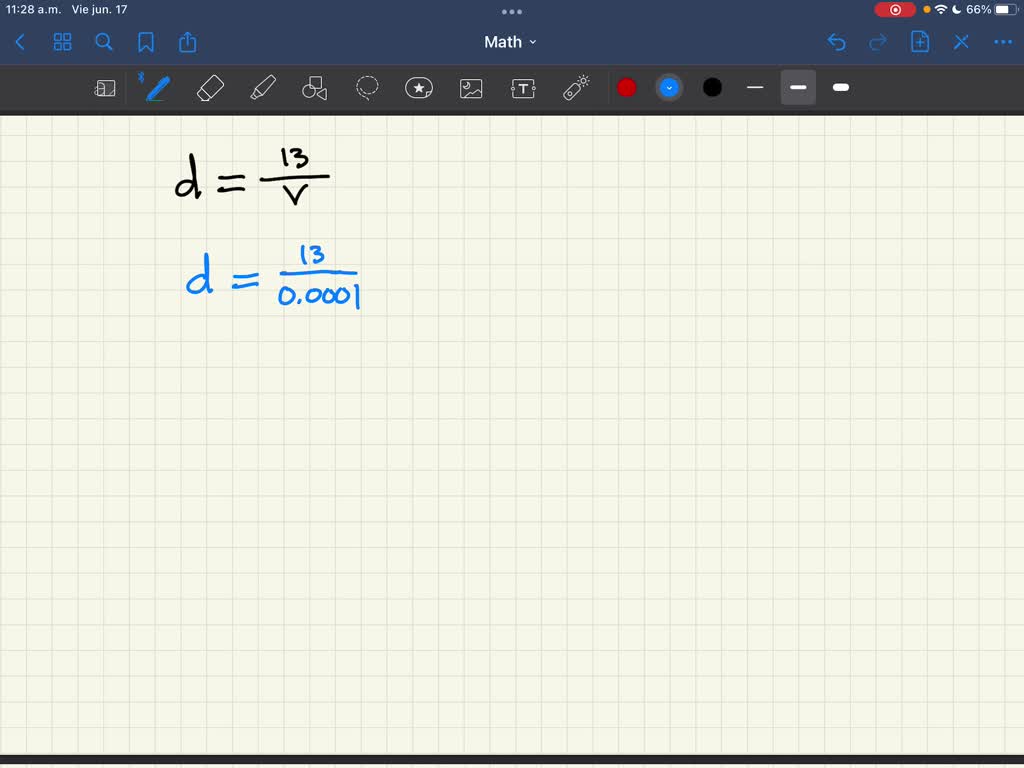
SOLVED: The equation d=13/v shows that the density of a particular substance equals a mass of 13 grams divided by the volume of the substance. What happens to the density as the
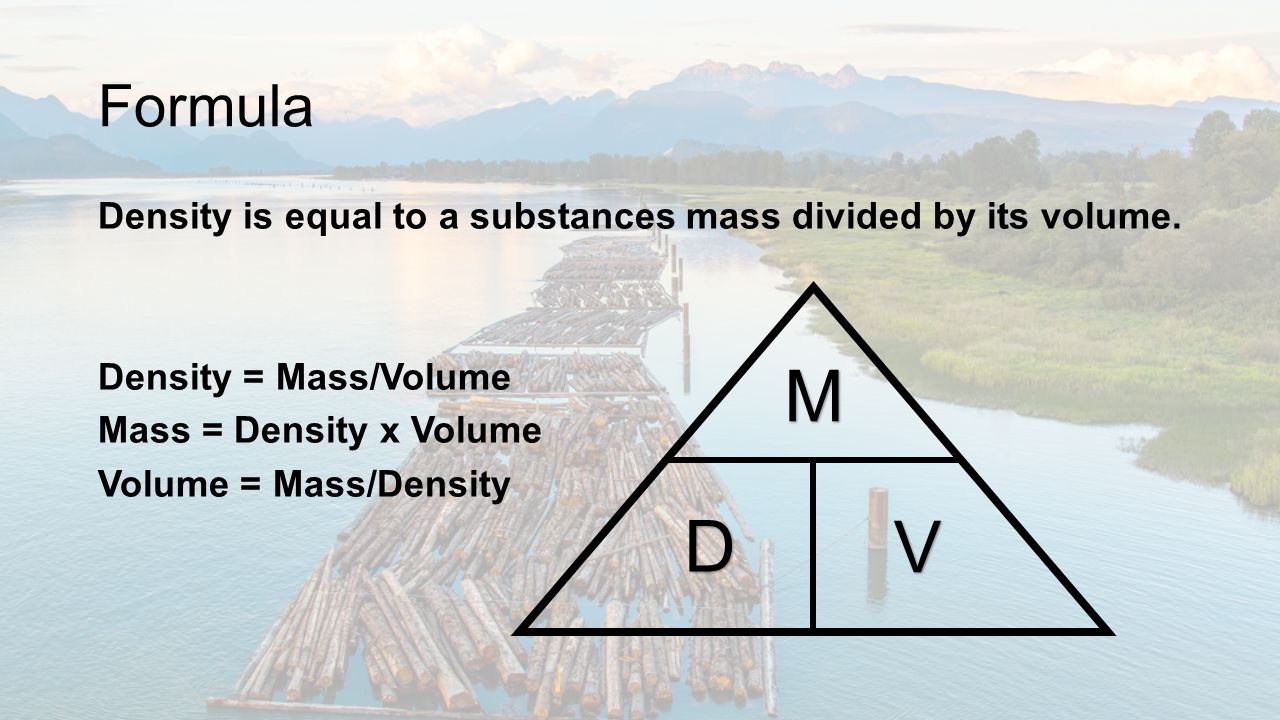
What is Density? Formula Density is equal to a substances mass divided by its volume. Density = Mass/Volume Mass = Density x Volume Volume = Mass/ Density. - ppt download